

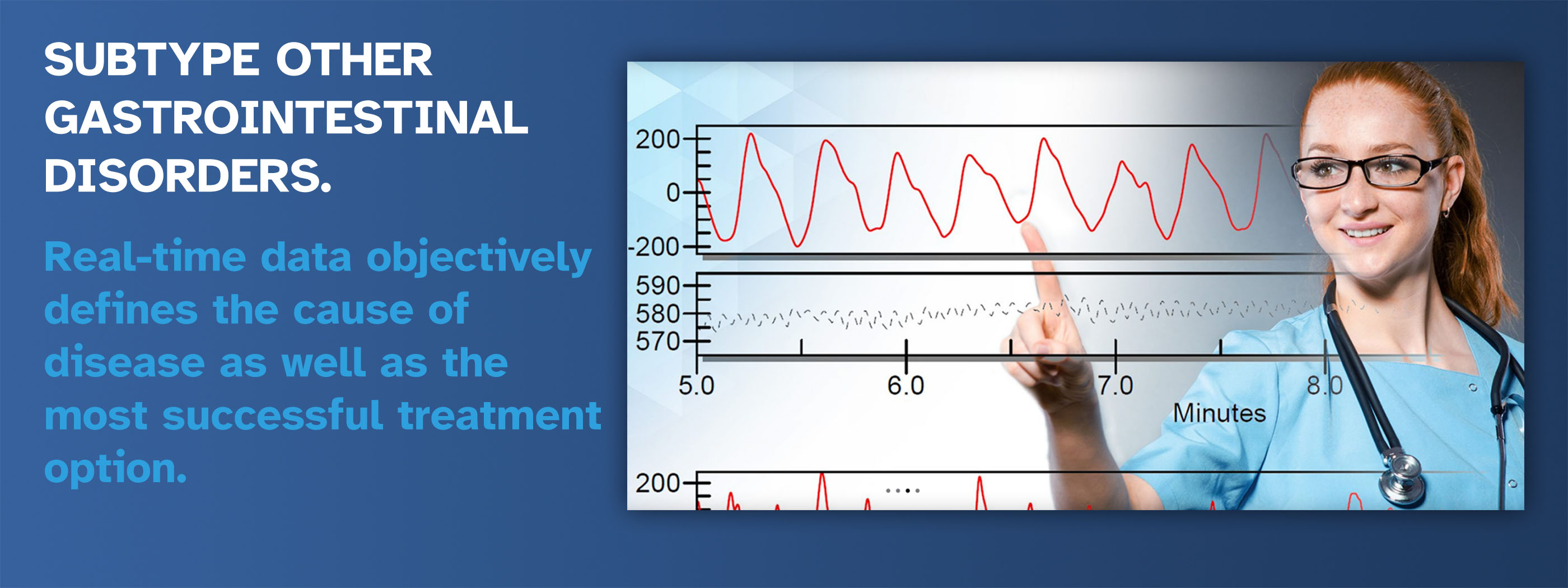

Next Generation Diagnostics for Gastrointestinal Disease
Building upon 30 years of advances in artificial intelligence, device development and medical simulation derived hardware and software integration, the 3CPM Company pioneered future advancements in Electrogastrography.
Miniaturization of hardware components coupled with evidence-based AI-driven software diagnostics and interpretation gave rise to the next generation technology which serves as the basis for current and developing platforms in Electrogastrography.
Leading the way forward is 3CPM’s proprietary matured technology, completely automated signal acquisition and AI based automation of diagnostic and recommended therapeutic applications.
Electrogastrography EGG
- ONLY APPROVED DEVICE
- ACCURATELY CHARACTERIZE GASTROINTESTINAL MOTILITY DISORDERS
- GASTROPARESIS
- FUNCTIONAL DYSPESIA
Features
- Non-Invasive, simple 45-minute test.
- AI derived patented technology.
- Subtype disease to optimize treatment.
- Personalize diagnosis and therapy.
Attributes:
- Non-invasive and very simple to perform 45 minute test
- Patented breakthrough technology
- Personalized diagnosis of gastrointestinal disorders
- Subtyping myoelectrical activity to optimize treatment outcomes
- Defining pathways to control or cure previously uncurable illnesses
- Accurate, Timely and Objective diagnosis of:
- dyspepsia
- gastroparesis
- reflux
- motility disorder
- pyloric dysfunction
- nausea
- vomiting
- bloating
- abdominal distension
- abdominal pain


